Glass is not a single, uniform material, and assuming otherwise is one of the most costly misconceptions in medical device engineering. When your design brief calls for a component with specific optical transmission or hydrolytic resistance, the terminology you use shapes every decision that follows, from material selection to supplier dialogue to regulatory submission. Misapplied terms create ambiguity in technical drawings, cause procurement errors, and can compromise device safety. This guide defines the essential vocabulary of medical glass, clarifies the distinctions between common glass types, and shows you how to apply that knowledge with confidence throughout your development process.
Table of Contents
- The role of specialised glass in medical devices
- Fundamental medical glass terms defined
- Comparing common glass types used in medical engineering
- How to apply glass terminology in the design process
- Why mastering glass terminology drives better device design
- Advance your medical device project with precision glass expertise
- Frequently asked questions
Key Takeaways
| Point | Details |
|---|---|
| Terminology accuracy | Clear understanding of glass terminology helps avoid costly design errors. |
| Material selection | Matching glass properties to device needs ensures performance and safety. |
| Effective communication | Fluent use of correct terms improves supplier and team collaboration. |
| Application in projects | Practical use of terminology streamlines design and procurement processes. |
The role of specialised glass in medical devices
Glass plays a foundational role across diagnostic, surgical, and analytical equipment. Endoscope lenses, microfluidic chips, laboratory slide covers, imaging windows, and syringe barrels all depend on glass components engineered to precise tolerances. The material is not chosen for convenience. It is chosen because it offers a combination of properties that polymers and metals simply cannot replicate at the performance levels medical applications demand.
The four properties that most frequently drive glass selection in medical engineering are:
- Biocompatibility: The material must not provoke adverse biological reactions when in contact with tissue or fluids.
- Thermal resistance: Components must withstand sterilisation cycles, including autoclaving at 121°C or higher, without distortion or cracking.
- Optical clarity: Diagnostic and imaging devices require consistent light transmission across defined wavelength ranges.
- Chemical inertness: Glass must resist degradation when exposed to reagents, cleaning agents, and bodily fluids.
Three glass families dominate medical device applications. Borosilicate glass, which contains boron trioxide, offers excellent thermal resistance and chemical durability, making it the standard choice for laboratory glassware and pharmaceutical packaging. Aluminosilicate glass incorporates aluminium oxide to achieve superior hardness and scratch resistance, suited to touchscreen interfaces and protective covers on diagnostic instruments. Fused silica, essentially pure silicon dioxide, delivers the highest optical transmission across ultraviolet, visible, and infrared wavelengths, making it indispensable in spectroscopy and laser-based surgical tools.
Material choice is not a secondary consideration. It determines whether a device performs reliably across its entire service life.
Specialised glass types are chosen for their unique chemical and mechanical properties relevant to medical applications. Selecting the wrong type, even when dimensions are correct, can introduce failure modes that only appear under real-world operating conditions. Exploring the full range of technical glass options available for medical use is the first step towards confident specification. For devices where vision and light transmission are critical, understanding the performance envelope of optical components is equally important.
With the landscape set, let’s demystify the types and underlying properties of medical glass.
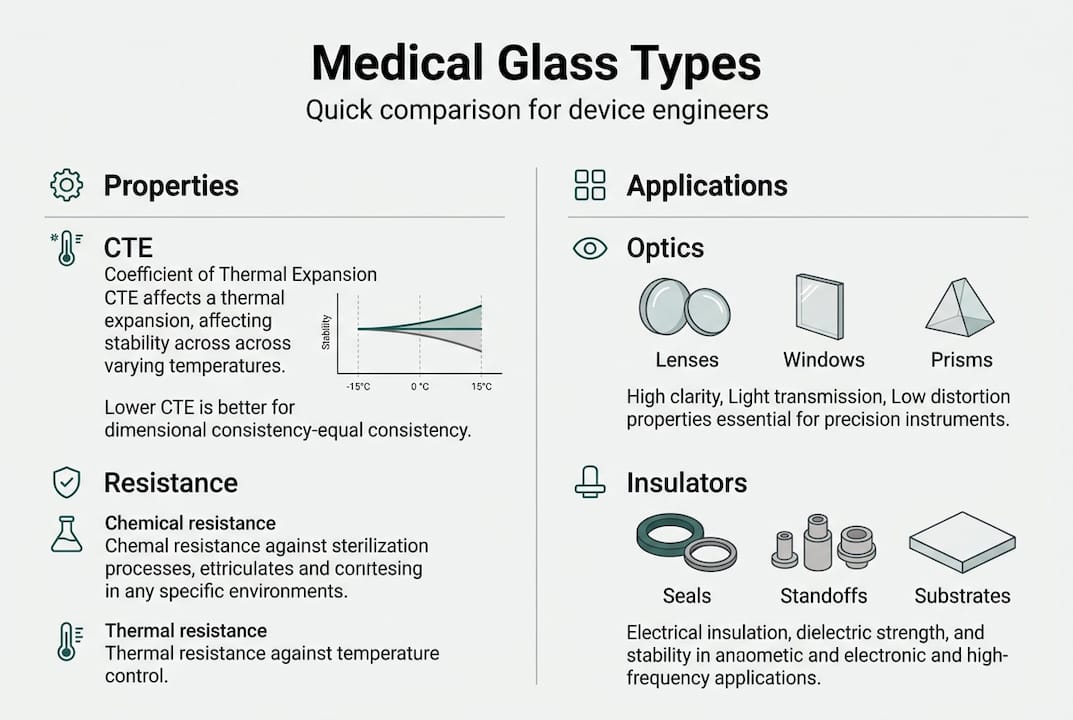
Fundamental medical glass terms defined
Now that we understand the purpose of glass in medical devices, it is essential to speak the same technical language. Misaligned vocabulary between design teams, procurement, and glass manufacturers is a persistent source of error. The following terms appear repeatedly in specifications, standards, and supplier dialogues.
- Annealing: A controlled slow-cooling process applied after forming. It relieves internal stresses that would otherwise cause spontaneous cracking. Annealed glass is dimensionally stable and easier to cut or grind to final tolerances.
- Coefficient of thermal expansion (CTE): The rate at which glass expands per degree of temperature increase, expressed in parts per million per degree Celsius. Matching CTE between glass and adjacent materials prevents stress fractures at joints.
- Hydrolytic resistance: A measure of how resistant glass is to attack by water. Classified under ISO 719 and ISO 720, it is critical for any component in contact with aqueous solutions or subject to steam sterilisation.
- Optical transmission: The percentage of incident light that passes through a glass component at a given wavelength. Stated as a spectral curve, not a single figure.
- Devitrification: The undesirable crystallisation of glass during processing or use. It reduces optical clarity and mechanical integrity, and is a particular risk when glass is held at elevated temperatures for extended periods.
| Term | Unit or classification | Relevance to device design |
|---|---|---|
| CTE | ppm/°C | Material compatibility at joints |
| Hydrolytic resistance | ISO class 1, 2, or 3 | Sterilisation and fluid contact |
| Optical transmission | % per wavelength (nm) | Imaging and diagnostic accuracy |
| Devitrification | Visual or microscopic inspection | Processing and long-term stability |
Understanding terms like annealing and coefficient of expansion is critical for manufacturing success. Without this fluency, even well-intentioned specifications can produce components that fail qualification testing. Our range of medical glass products is manufactured with each of these parameters documented and traceable.

Pro Tip: Pay special attention to CTE when designing assemblies that join glass to metal or ceramic housings. A mismatch of even a few ppm/°C can generate sufficient stress during sterilisation cycles to initiate micro-cracking that is invisible until the device fails in service.
Armed with the terminology and glass types, let’s explore how to put this knowledge into practice.
Comparing common glass types used in medical engineering
With foundational terms covered, let’s compare how those characteristics differ across glass types. Understanding these distinctions prevents over-specification in some cases and dangerous under-specification in others.
| Glass type | CTE (ppm/°C) | Max service temp (°C) | Chemical resistance | Typical medical application |
|---|---|---|---|---|
| Borosilicate | 3.3 | 500 | Excellent | Lab glassware, syringe barrels, sight glasses |
| Aluminosilicate | 4.0 | 700 | Very good | Instrument covers, touchscreen panels |
| Fused silica | 0.55 | 1000+ | Outstanding | UV optics, laser windows, spectroscopy |
| Soda-lime | 9.0 | 300 | Moderate | Microscope slides, low-cost covers |
Borosilicate and aluminosilicate glass differ significantly in chemical durability and thermal behaviour. Borosilicate remains the workhorse of medical glass due to its balance of cost, processability, and performance. Aluminosilicate offers greater hardness but is more demanding to fabricate to tight tolerances.
Key considerations when selecting between these types:
- Miniaturisation: As devices shrink, thinner glass walls are required. Fused silica and aluminosilicate tolerate tighter tolerances without edge chipping.
- Imaging performance: Soda-lime glass absorbs UV wavelengths, making it unsuitable for fluorescence-based diagnostics. Fused silica or UV-grade borosilicate must be specified instead.
- Sterilisation compatibility: Repeated autoclaving cycles test hydrolytic resistance. Borosilicate Type I (ISO class 1) is the minimum standard for components in direct fluid contact.
Specifying the glass type by name alone is insufficient. Always state the relevant standard, grade, and performance class to eliminate ambiguity.
Recent advances in thin-film coatings and precision forming have expanded what each glass type can achieve. Anti-reflective coatings on borosilicate, for instance, now achieve transmission values above 99.5% in the visible range. Reviewing the full catalogue of glass varieties available for medical engineering reveals how far material performance has advanced in recent years.
How to apply glass terminology in the design process
Armed with the terminology and glass types, let’s explore how to put this knowledge into practice across your development workflow.
- Technical drawings: State the glass type, grade, and applicable standard on every drawing. Include CTE, surface flatness, and optical transmission requirements as explicit tolerances, not narrative notes.
- Requests for quotation (RFQs): Use standardised terminology consistently. Suppliers interpret ambiguous language differently, and a phrase like “optical quality” without a transmission specification will yield inconsistent quotes and inconsistent parts.
- Design review meetings: Establish a shared glossary at the start of any multi-disciplinary project. When mechanical engineers, optical designers, and procurement teams use the same definitions, review cycles shorten significantly.
- Supplier qualification: Ask suppliers to confirm their interpretation of key terms in writing. If their definition of “annealed” differs from yours, that discrepancy will surface in the component, not in the conversation.
- Regulatory submissions: Regulatory bodies expect precise material characterisation. Vague glass descriptions in technical files are a common cause of requests for additional information, which delay market access.
Effective use of terminology improves supplier communication and reduces costly production errors. This is not a theoretical benefit. It translates directly into fewer non-conformances, shorter lead times, and lower total cost of ownership. Our glass engineering services are structured to support teams at every stage of this process, from early specification through to final qualification.
Pro Tip: Document agreed-upon definitions in your project specification document and reference them in every supplier communication. This single practice eliminates a significant proportion of the ambiguity-driven rework we observe in complex device programmes.
Why mastering glass terminology drives better device design
We have worked with enough development teams to observe a consistent pattern. Projects that struggle with glass-related failures rarely have the wrong glass. They have the right glass specified imprecisely. Generic terms like “toughened” or “optical grade” without accompanying standards create gaps that manufacturing tolerances cannot bridge.
The vocabulary of glass is also evolving. As medical devices incorporate thinner substrates, curved optics, and integrated photonic functions, new terms are entering common use. Engineers who stay current with this language are better positioned to evaluate emerging materials and communicate requirements to specialist manufacturers without translation errors.
Our view is straightforward. Investing time in precise glass terminology is not administrative overhead. It is a risk reduction strategy. The cost of a terminology-driven failure, whether a failed sterilisation validation or a rejected regulatory submission, far exceeds the time spent building a shared glossary at project outset. Accessing reliable specialised glass knowledge early in development is one of the most practical steps a device team can take.
Advance your medical device project with precision glass expertise
Understanding glass terminology is the foundation. Translating that understanding into a reliable, compliant component requires a manufacturing partner with the depth of expertise to match your specifications precisely.

At Glass Precision, we support medical device manufacturers and engineers with custom technical glass for medical applications, from initial specification review through to precision fabrication and quality-assured delivery. Whether you are sourcing borosilicate sight glasses, fused silica optical windows, or aluminosilicate instrument covers, our team is ready to work with you. Explore our full range of precision glass solutions or contact us to discuss your project requirements with a specialist.
Frequently asked questions
What is the difference between annealed glass and tempered glass in medical devices?
Annealed glass is slowly cooled to relieve internal stress and produce a uniform structure, while tempered glass is heat-treated to increase strength and produce a safer fragmentation pattern on breakage. Annealed and tempered glass have distinct thermal processing methods that affect both mechanical strength and in-service behaviour.
Why is hydrolytic resistance important for medical glass?
High hydrolytic resistance prevents glass from leaching ions or degrading when in prolonged contact with water, reagents, or bodily fluids, preserving both device sterility and long-term durability. Hydrolytic resistance is a key factor in medical glass selection for sterility and chemical stability.
How can incorrect terminology affect device manufacturing?
Misused or ambiguous terms cause material mismatches between specification and supply, leading to production delays, failed qualification testing, and compromised device performance. Miscommunication in material specification leads to increased risk and cost across the development programme.
What is the coefficient of expansion in relation to glass?
The coefficient of thermal expansion describes the rate at which glass expands per unit temperature rise, and it must be matched carefully to adjacent materials to prevent stress-induced cracking at joints or interfaces. The coefficient of expansion must be considered when joining glass to other device materials during assembly and sterilisation cycles.





One Response
https://shorturl.fm/kufS6